Regulatory Overview
Because of the tremendous advances in gene editing in agriculture there is a growing consensus among policy makers to consider and regulate gene edited crop varieties in the same way as conventional varieties when the genetic changes are similar to those that occur in conventional breeding or nature.
A major driver of these efforts is the need to harmonize globally the regulatory requirements for trait development using gene editing to address the massive productivity challenges in farming such as disease, insects, drought and fertilizer use in the face of climate change.
Background and Status
The goal of these global efforts to harmonize regulation associated with gene editing is to fix the patchwork of uncertain and complex regulations promulgated to ban or limit GMO’s or the use of foreign DNA in breeding traits. They are addressing a global understanding that the lack of regulatory clarity, including consistent and predictable legislation with clear technical criteria, has stifled innovation and the realization of the full potential of these technologies.
Technology breakthroughs driving these proposed regulatory changes are the new gene editing technologies that mimic the process by which genomic change occurs in nature. Because gene-edited plant traits are indistinguishable from those that arise in nature the proposed regulations would treat these traits on the same basis as traits produced by conventional breeding. The regulations for traits that are developed using conventional breeding are globally harmonized. These new proposed changes would, in essence, have gene-edited traits logically follow those same regulations.
Agriculture is one of the rare global systems where global regulatory policy can directly address both carbon footprint, and impact on the environment, as well as sustainability, productivity, and profitability of the global food supply. These new innovations from biological crop protection will enable the farmer to better adapt to their rapidly changing environment, to lower their costs and to materially improve their workplace and working conditions. These are the real drivers in this push for a new regulatory environment to enable a new era of growth and innovation in global crop farming practice. As Copa-Cogeco, a group that represents 22 million European farmers, put it, “[these technologies] are not a luxury but an urgent necessity for the vitality of the whole EU farming model…”
Advances in Global Regulatory Policy
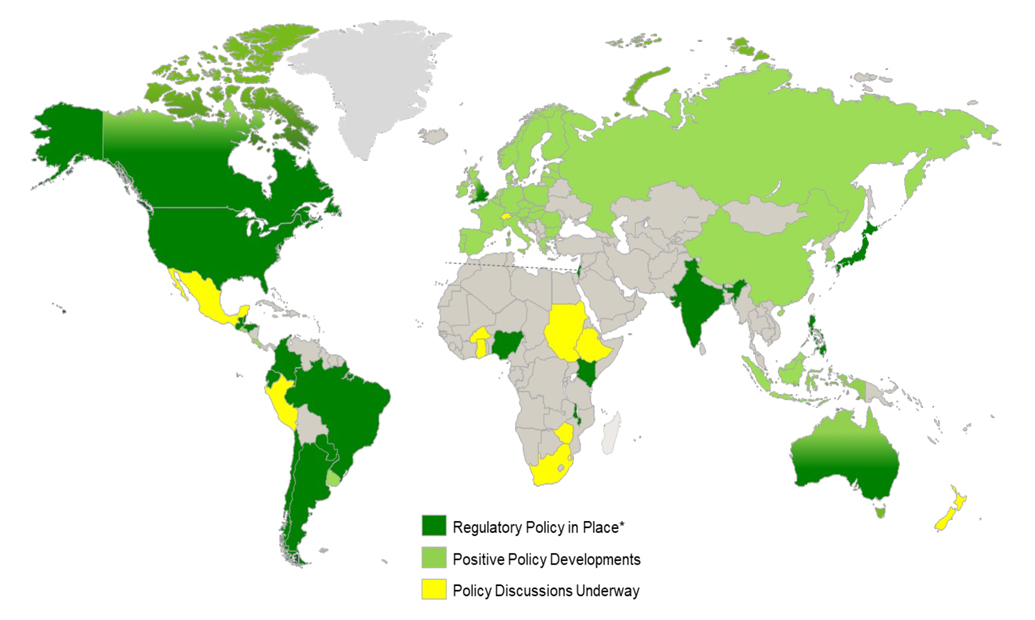
The map below attempts to show the progress region-by-region of the status of regulatory efforts associated with precision gene editing. Dark Green are the geographic areas, including the United States and much of South America, in which countries have adopted policies to regulated gene editing like conventional breeding. Light Green shows geographic areas where policy developments are underway—in most of these areas, such as the European Union, traits from gene-editing are currently regulated as GMO. Policy developments underway means that these countries are developing new regulations to regulate traits developed from gene editing like traits developed from conventional breeding. Yellow means policy discussions are under way.